 Many chemical and physical properties of elements are not easy to understand, and many properties are related to each other, making it complex to understand the periodic table and its elements. So let’s discuss the reasons behind student’s difficulty understanding this topic: Rather it is kind of boring and hectic topic for students. Why can the periodic table and its principle be tricky to understand?Īlthough it seems very interesting to look at the periodic table to understand it, it is not an interesting topic for the students. The elements in the periodic table are classified as metals, non-metals, metalloids, halogens, noble gases, and rare earth elements. If you want to put an element in the periodic table, you would notice its flame color and then put it in the respective row and column. It is one of the parameters of the classification of the periodic table. Since 1961, the standard unit of atomic mass has been one-twelfth the mass of the isotope of carbon-12.Įlements are also classified by flame color test. It is the ratio of a chemical element’s atoms to some standard. 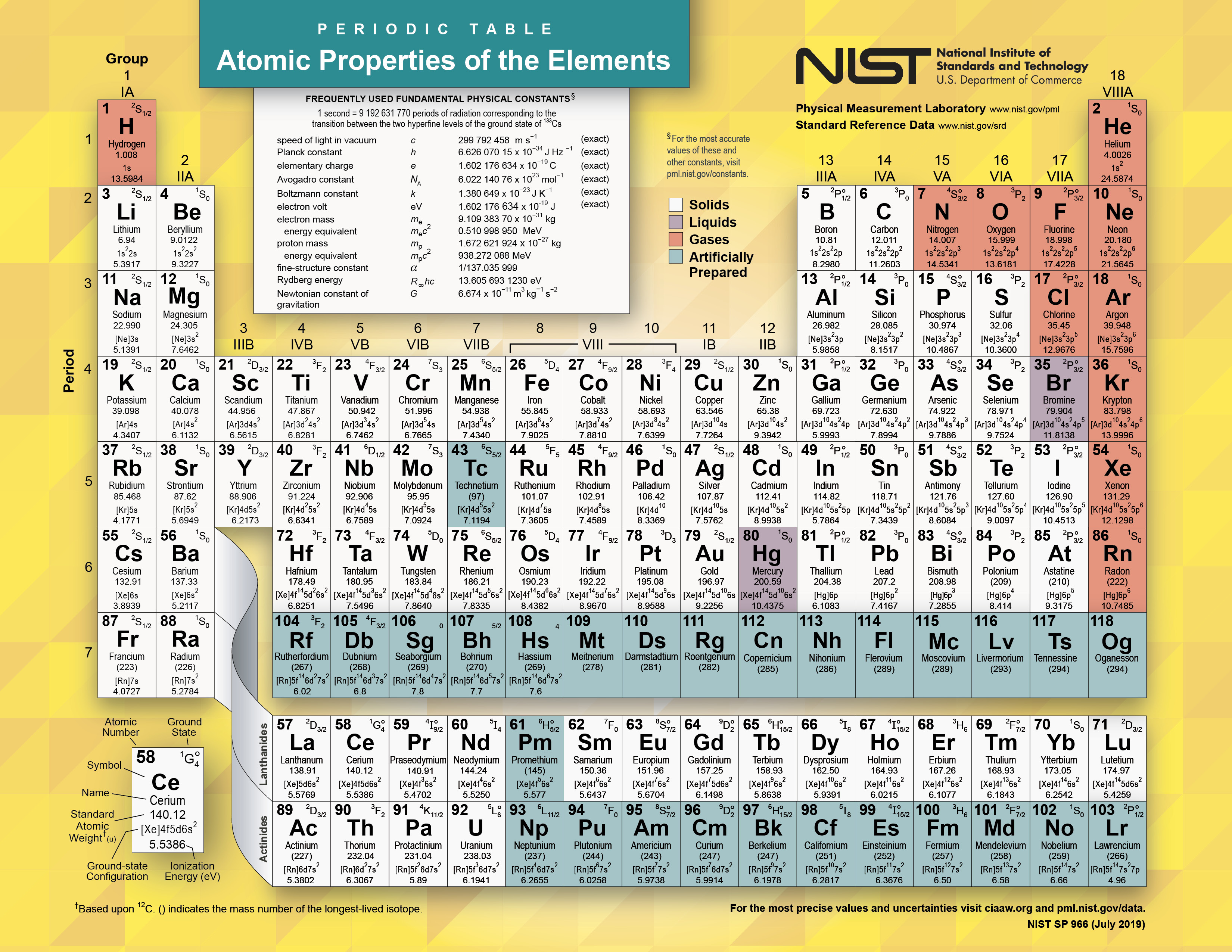
Atomic weight is also known as relative atomic mass. It means that elements' chemical and physical properties reoccur periodically if the elements are arranged according to their atomic weights. This is the basic principle behind the existence of the periodic table. The periodic table is based upon the principle that states, “the properties are a periodic function of their atomic mass”. They can easily make chemical bonds with valence electrons of other elements. These electrons are known as valence electrons. Meanwhile, the group shows the number of electrons in an atom’s outermost shell. In a single row, all elements have the same number of atomic orbitals (spaces where electrons exist). There are seven rows and 18 columns in the periodic table. The rows in a periodic table are known as periods, and each column is known as a group. The periodic table is organized according to atomic weight and valence electrons in the outer shells of elements. Based on these properties, elements are divided into different categories. The physical properties of periodic table elements are color, luster, malleability, hardness, solubility, and electrical conductivity. In a periodic table, elements are arranged in different rows and columns according to their physical and chemical properties.Ĭhemical properties that distinguish elements from each other are atomic number, atomic mass, electronegativity, density, melting point, boiling point, and ionic radius. 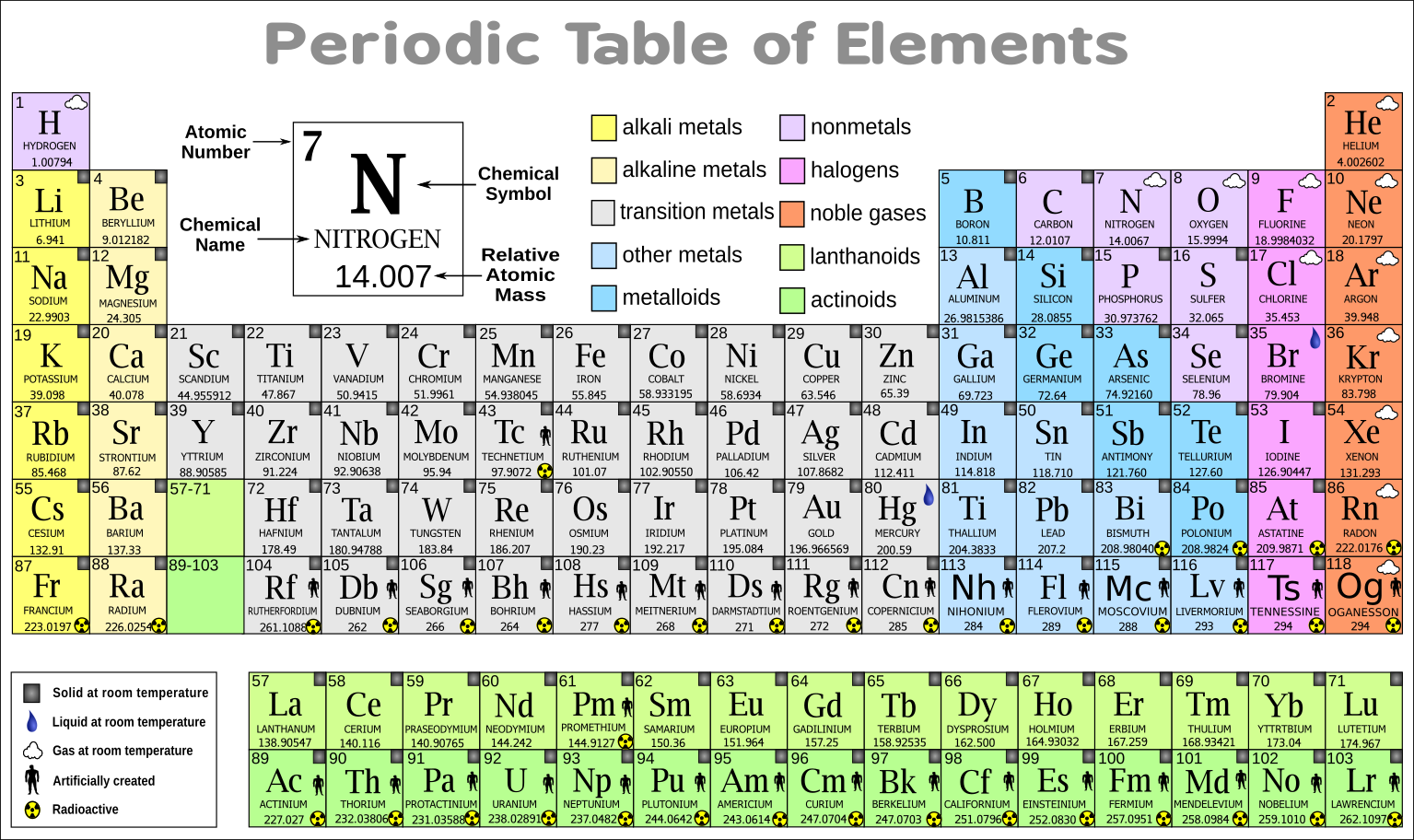
Looking at carbon, for example, its symbol (C) and name appear, as well as its atomic number of six (in the upper left-hand corner) and its atomic mass of 12.11.The periodic table is an arrangement of elements according to their chemical and physical properties. In addition to providing the atomic number for each element, the periodic table also displays the element’s atomic mass. In the periodic table, shown in Figure 1, the elements are organized and displayed according to their atomic number and are arranged in a series of rows and columns based on shared chemical and physical properties. Elements also have specific chemical reactivity, the ability to combine and to chemically bond with each other. The properties of elements are responsible for their physical state at room temperature: they may be gases, solids, or liquids. Devised by Russian chemist Dmitri Mendeleev (1834–1907) in 1869, the table groups elements that, although unique, share certain chemical properties with other elements. The different elements are organized and displayed in the periodic table. Understand the periodic table of elements and how to use it to understand elements. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
